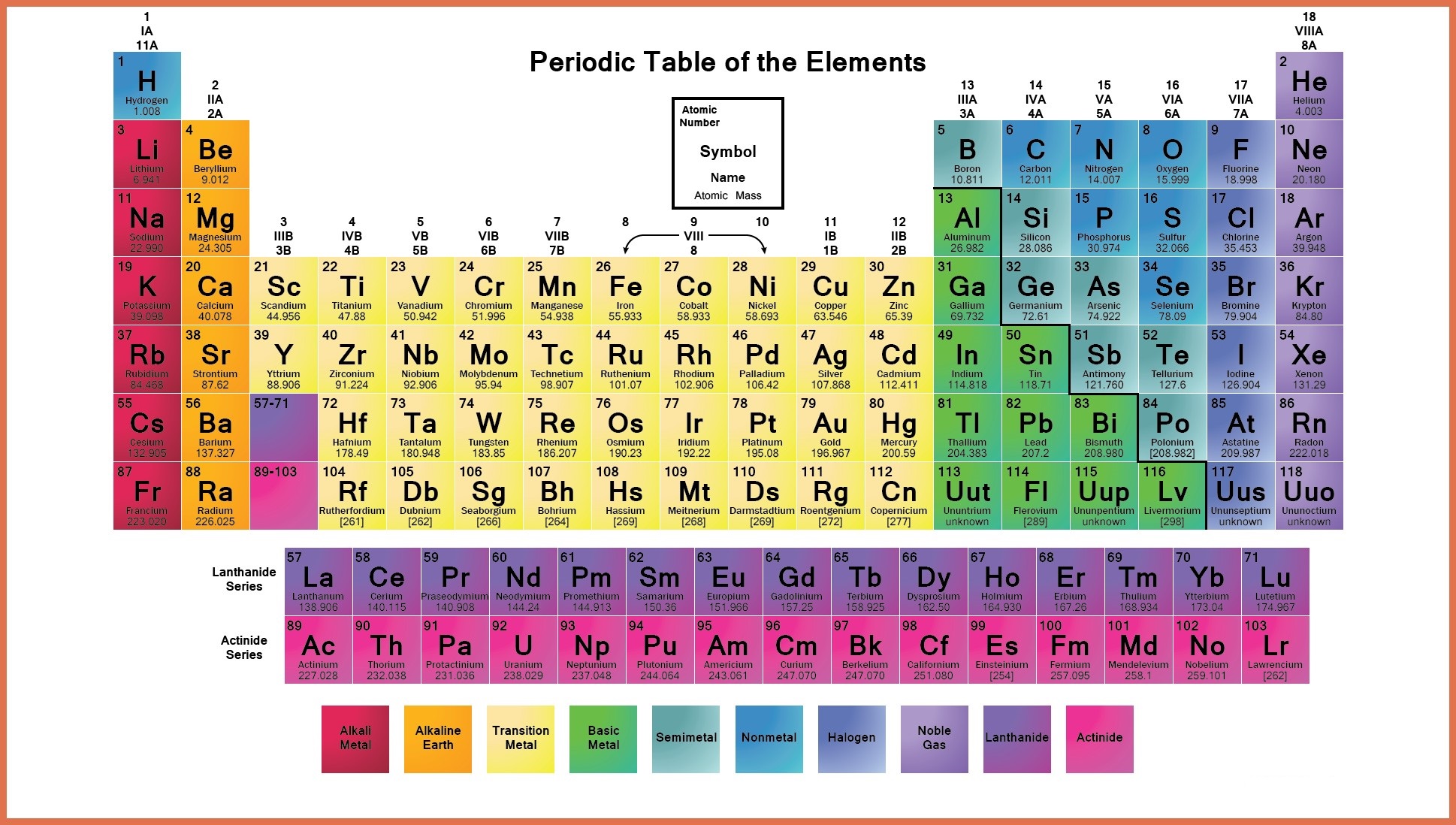 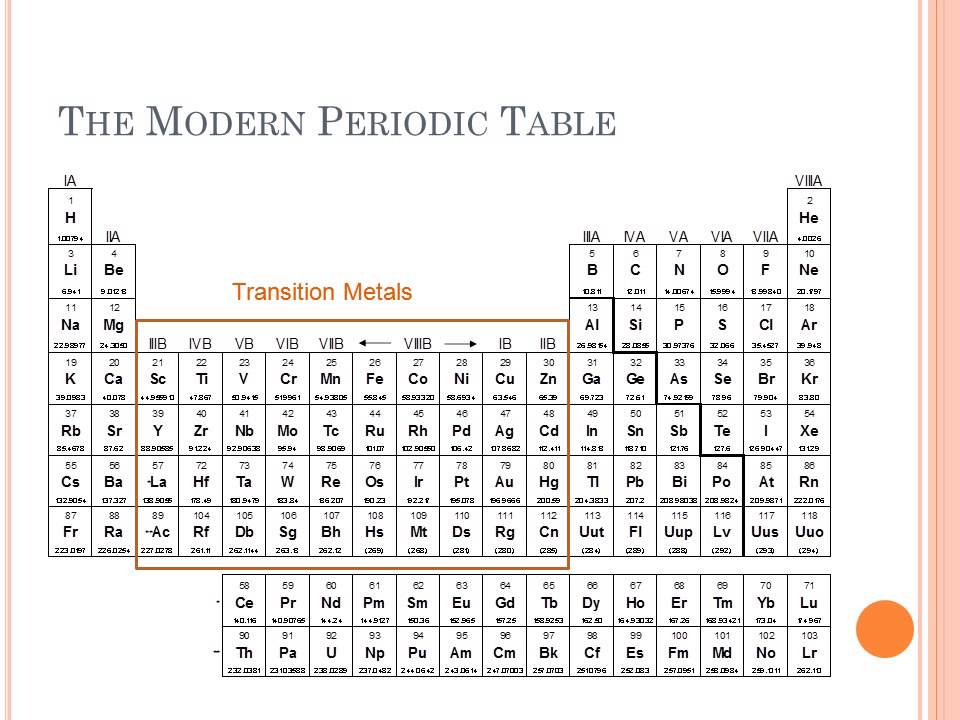
To be more concrete, the ionic radius of an atom is defined as half the distance between ions that are just barely contacting each other. What is the ionic radius exactly? As a quick definition, an atom’s ionic radius is a measurement of the ions taken as the atom is positioned inside of the crystal lattice. 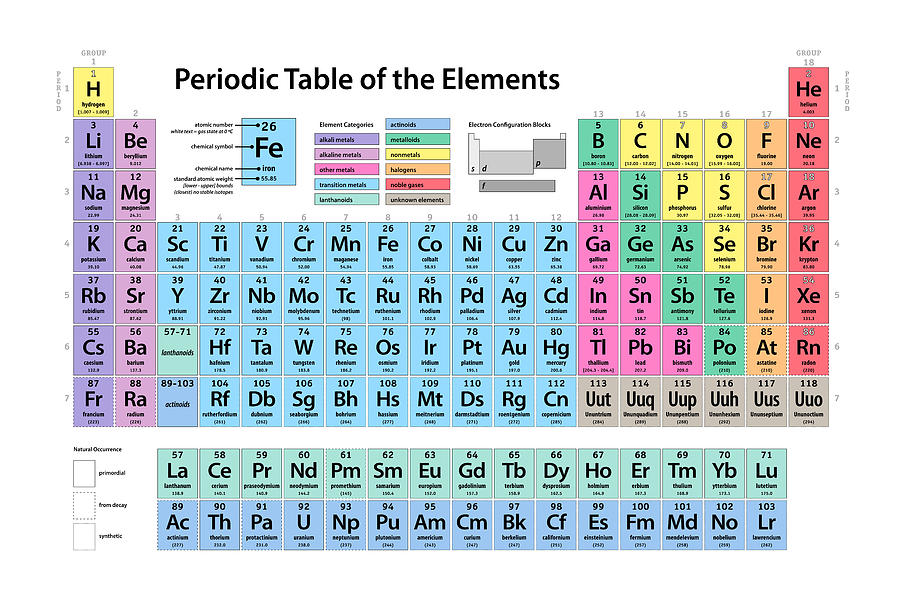
Photo: Photo: () by Geralt via Pixabay, Pixabay License () Ionic radius tends to decrease as one reads across the table from left to right. The ionic radius trend describes how as one follows the periodic table from top to bottom, the ionic radius of the elements within the table tends to increase. Elements found within the same column have electron distributions that are identical, and because of this, they have very similar chemical properties and reactions. An element’s period number represents the highest energy level that an electron in that element possesses. Rows on the periodic table are referred to as periods, while the columns on the periodic table are referred to as groups. In this article, you will find a labeled periodic table of the elements, as well as information about the trends relating to the periodic table, or the patterns you can use to better understand the table. The elements on the periodic table run from top to bottom and left to right in order of increasing atomic number, and in general the order of the elements is correlated with their atomic mass. The periodic table of the elements is a representation of all of the chemical elements that have been discovered.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
